Development
CANDO kicked off in August 2014. Over seven years the project will progress through several phases. Initial phases focus on technology design and development, followed by rigorous testing of performance and safety. The aim is to create a first-in-human-trial in the seventh year.
The project has recently passed its third milestone in the project, coinciding with the third year of the project. Significant progress has been made in identifying the optimal design for elements of the device and selection of a number of potential vectors for delivering the gene therapy. We are now able to move forward with testing prototypes of the device and the gene therapy for safety and efficacy in advance of applying for ethical and regulatory approval for a human trial.
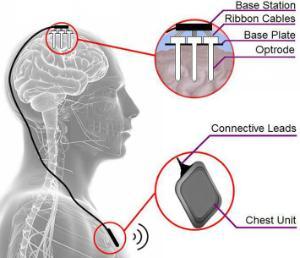
We are now in the position to be able to start reporting findings at conferences, symposiums and in papers. Click on the link on the left to find out more about our findings.
